In a new publication, Mayo researchers and collaborators report a previously unknown interaction between adenovirus vectored vaccines and a protein released by platelets that may provide a way to engineer improved viral-vector-based vaccines in the future. The discovery science work, which is published in Science Advances, investigates extremely rare blood clotting events observed in patients after vaccination with an adenovirus-based COVID-19 vaccine.
To the researchers, the side effect, called "thrombosis with thrombocytopenia syndrome," also known as "vaccine-induced thrombotic thrombocytopenia," seemed similar to a rare condition called "heparin-induced thrombocytopenia." That condition is a drug reaction to the blood thinner heparin. It is caused by an immune reaction that directs platelets to clot when heparin is in the bloodstream. Previous work on viral-vector-based therapies link decreased efficacy to antibodies to the virus if a patient has previously been exposed to the vector, and to interaction of the vaccine with parts of the blood clotting pathway, such as platelets.
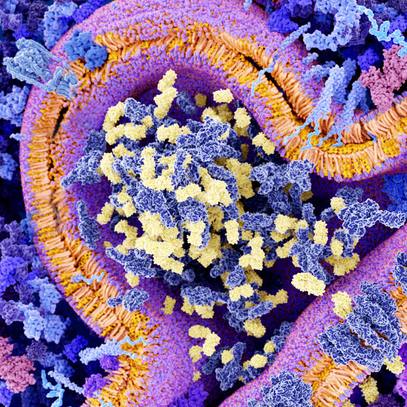
To explore the idea of a vector-blood clotting interaction, the team ― assembled from researchers at Mayo Clinic, Arizona State University, Michigan State University, and Cardiff University, among others ― first imaged the adenovirus vector. In a technique called "cryo-electron microscopy," viral particles were frozen and then imaged using a beam of electrons. Since the particles are oriented in different ways, a computer program evaluates all the options to create a 3D structure, a detailed view of the topography of the viral "shell" called the capsid.
With the structure of the capsid clarified at the highest resolution to date, the authors identified the main attachment protein of the virus.

They then tested other proteins that could interact with the adenovirus. In cell culture models, and by using a technique to measure binding between the virus and receptor, the authors determined the adenovirus interacts with a protein called platelet factor 4. It is an immune-signaling protein, released by platelets when they are directed to gather and form a clot.
In the cell studies, the authors showed that a complex could be formed between the adenovirus-vector, PF4, and antibodies to PF4 , which the authors hypothesize might explain the rare thrombosis with thrombocytopenia syndrome recorded after adenovirus-based vaccine administration. They further suggest that altering the electric charge of the adenovirus vector might help prevent the syndrome, and make the adenovirus look different to similar viruses. That could prevent the immune system from neutralizing the virus before it can have a therapeutic effect.
The next steps for the team are to clarify how this interaction between the adenovirus vector and PF4 may connect to the clinical symptoms experienced by the rare subset of those receiving a viral-vector-based vaccine.
For a full list of authors, including Mayo Clinic authors Alexander Baker, Ph.D., and Mitesh Borad, M.D., as well as funders and competing interest disclosures, see the paper in Science Advances.